 
Hence, correct order is fcc (74%) > bcc (68%) > simple cubic (52%). Packing efficiencies of different cells are shown in the above table. What is the correct order of packing efficiencies of different cells? Packing fraction of different types of packing in unit cells is calculated below: Hexagonal close packing (hcp) and cubic close packing (ccp) have same packing efficiency. 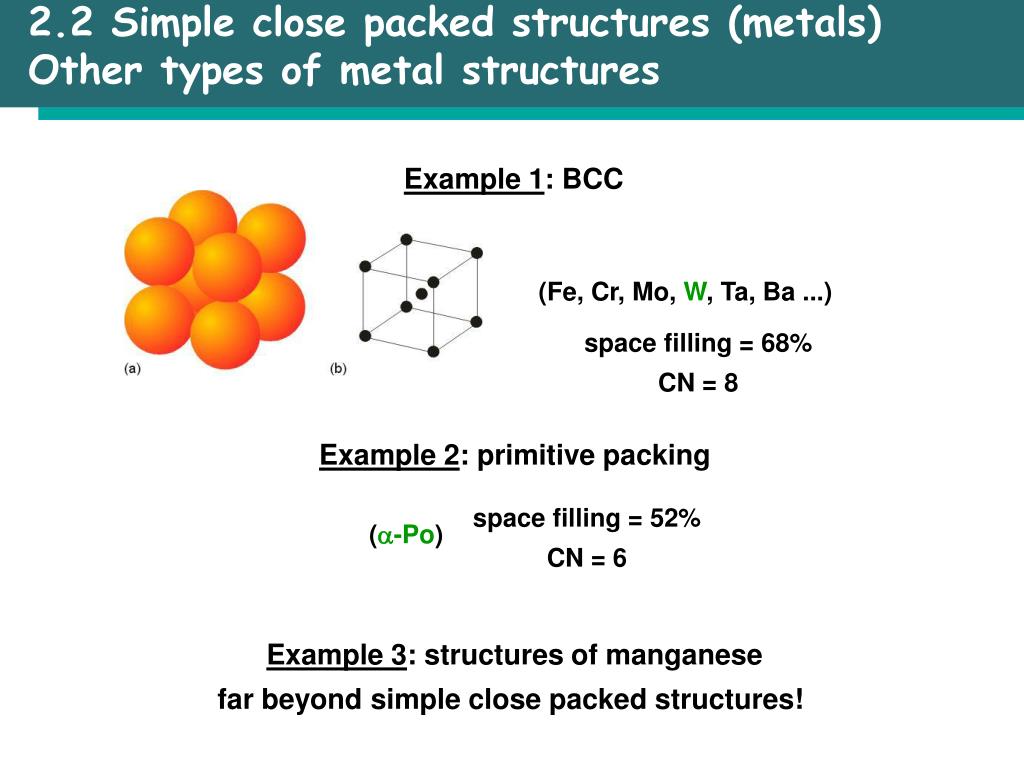
In this section we shall learn about packing efficiency. 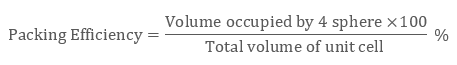
Percentage of spaces filled by the particles in the unit cell is known as the packing fraction of the unit cell. The packing efficiency of the simple cubic cell is 52. of effective atoms in hcp unit cell (Z) 6 Molar mass of A: Click to see the answer Q: Metallic iridium crystallizes in a face-centered cubic lattice, with one Ir atom per lattice point. In crystallography, atomic packaging factor (APF), packaging performance or packing fraction is definitely the small percentage of volume in a crystal construction that is occupied by constituent particles. Since a simple cubic unit cell contains only 1 atom. A: Cobalt crystallize in a HCP unit cell No. The HCP unit cell is of hexagonal geometry and is generated by the stacking of close-packed planes of atoms. What is the packing efficiency of a simple cubic cell? The unit cell that has the minimum or lowest Packing Efficiency is Simple Cubic with 52. Which unit cell has the minimum or lowest packing efficiency? Packing Fraction Formula = Volume Occupied by all constituent particles Total Volume of Unit CellUnit Cell Packing Efficiency – VEDANTUwww. 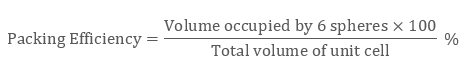
Packing Efficiency Formula = No of Atoms x Volume Occupied by 1 atom Total Volume of Unit Cell X 100 We can say that Packing Fraction, when multiplied by 100, is seen as a percentage, it becomes the Packing Efficiency of that particular cell. The volume of each atom is (4R3) and the volume of the unit cell is. The packing efficiency of simple cubic unit cell is 52. After reaching a yield stress at t x 180, t y 110 ( reduced units ), we observe a sudden increase of the amount of hcp structure as demonstrated in Fig. The packing factor is the fraction of space occupied by atoms, assuming that atoms are hard spheres Packing factor Examplele 1.3 Calculate the packing factor for the FCC cell There are 4 lattice points per cell and one atom per unit cell giving total 4 atoms per unit cell. The packing efficiency of Bcc unit cell is 68%. Thus, 68 of the total volume in bcc unit lattice is occupied by atoms and 32 is empty space or void volume. The packing efficiency of Fcc unit cell is 74%. The packing efficiency is the percentage of total space occupied by the particles. What is the packing efficiency of simple cubic unit cell? The packing efficiency of simple cubic unit cell is 52. The packing efficiency of Bcc unit cell is 68%. What is the packing efficiency of BCC and CCP? The volume occupied by 4 atoms is 4Ã-34‹Ïr3=316‹Ïr3 The packing efficiency =total volume of unit cellVolume occupied by atoms in a unit cell ‹Ã-100 The packing efficiency =88‹r3316‹Ïr3‹Ã-100=74. Here 74 volume of unit cell of simple cube is occupied by atoms and remaining 26 volume is vacant. With this work, we also demonstrate the possibility of routine static high-pressure experiments with conventional bevelled diamond-anvil geometry in the 0.3–0.4 TPa regime.What is the packing efficiency of a CCP unit cell?Ī CCP unit cell has 4 atoms per unit cell. Volume occupied by the atoms per unit cell (v) Volume of unit cell for a cubic system (V) a 3 Hence packing factor is 0.74 which shows that there is much more close packing present in FCC than BCC. Here we confirm this observation with a synchrotron-based X-ray diffraction experiment carried out within a diamond-anvil cell and report indications of the onset of the transition towards a bcc structure at pressures beyond 320 GPa. The hcp phase was identified at 217 GPa in an experiment (Akahama et al., 2006), and the bcc phase has been recently confirmed in a dynamic ramp-compression experiment coupled with time-resolved X-ray diffraction (Polsin et al. A Simple Cubic Crystal contains only single atom and hence the Volume Occupied by atoms is given as: The Volume of Cube a3 i.e. The face-centered cubic (FCC) has a coordination number of 12 and contains 4 atoms per unit cell. The hexagonal closest packed (HCP) has a coordination number of 12 and contains 6 atoms per unit cell. For aluminium, first principle calculations have consistently predicted a phase transition sequence fcc–hcp–bcc in a pressure range below 0.5 TPa. Volume of HCP Unit Cell A unit cell is the smallest representation of an entire crystal. At extreme pressures, common materials are thus expected to transform into new dense phases with extremely compact atomic arrangements that may also have unusual physical properties. With the application of pressure, a material decreases in volume as described in its equation of state, which is governed by energy considerations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
